Deicing Facts
Different Salts
Those of us who live in the snow-belt are fairly familiar with rock salt. Large trucks distribute it on roadways to keep traffic flowing. I would curse the salt producers and the drivers of these trucks, not because of damage to roadways, but to my cars. Salts of just about any type are quite corrosive to metal. The chloride ion aggressively attacks poor quality steel. Many cars built in the '70s used poor quality steel, which made rusting body panels a nightmare. Cars built today seem to use better steel and steel that has corrosion resistant coatings that minimize salt damage. It is still a good idea to rinse off your car as soon as possible once it is coated with salt spray. Always rinse up under each fender and as much of the undercarriage as well.
A Safer Salt
Within the past few years a new deicing salt has been introduced to the USA. It is magnesium chloride. It joined the ranks of the other popular deicing salts: sodium chloride, calcium chloride and potassium chloride.
Magnesium chloride is attractive for several reasons. Testing conducted by the National Research Council's Strategic Highway Research Program showed that calcium chloride and sodium chloride caused lots more damage to concrete than did magnesium chloride. The sodium chloride - common rock salt - actually caused 63 times more damage than the magnesium chloride. This is a huge difference. The calcium chloride eroded 26 times more cement than the magnesium chloride. That is also a significant amount.
 Get a 24-page guide right now that answers all your questions about Deicing Salts. Will it RUIN your concrete? Did you know that salt can SERIOUSLY harm you? What about your expensive landscaping? You can have all these answers and more in less than a minute. Buy it NOW. Get a 24-page guide right now that answers all your questions about Deicing Salts. Will it RUIN your concrete? Did you know that salt can SERIOUSLY harm you? What about your expensive landscaping? You can have all these answers and more in less than a minute. Buy it NOW. |
Magnesium chloride is also less corrosive to metal such as tin, steel and aluminum (see Author's Note at the end of this column). The reason lies in the fact that it has less chlorides available than calcium chloride and sodium chloride. Remember, it is the chloride ion that attacks metals.
If you use significant amounts of salt or plan to, you need to paint all steel that is in contact with the concrete. This means that the steel bars or mesh used in concrete need to be primed with a rust inhibitive paint. Steel railing posts or aluminum posts need to be treated as well before they are cemented into place in a hole in a sidewalk or in a set of steps. Take this extra time and the corrosion will be minimal.
Plants, Animals & Carpets
Magnesium chloride is friendly to plants, streams, rivers and lakes. The primary reason for this is its lower chloride content. Magnesium itself is often found in many fertilizers.
Traditional deicing salts, like sodium and calcium chloride, leave white powder residue on carpets once they dry. Magnesium chloride doesn't do this.
Your pets should prefer magnesium chloride as well. It is not nearly as toxic as the other available deicing salts.
A Window of Vulnerability
Fresh concrete can be hurt by deicing salts if it is poured too late/early in the season. Concrete needs to be moist cured at or above 50 degrees F for a minimum of 7 days after it is poured. This curing controls and minimizes the release of water within the concrete.
Once cured, the concrete needs to dry for a minimum of 30 days before it is exposed to freeze-thaw conditions and deicing salts. Keep this in mind if you are pouring concrete early or late in the season!
Salt Toxicity
If you are not concerned with the environment, you should be. Each day there are more people on the planet. We need to adopt a sense of stewardship for future generations. You can do that by using products that are kind to the environment. Magnesium chloride is a deicing salt that is very non-toxic. In fact, it is the least toxic deicing salt - yet it is one of the most effective.
Look at the chart below to see how magnesium chloride - referred to as MAG - compares to other salts and some common household products you use and consume. Whatever you do, don't consume the top two items in the chart - cyanide and strychnine! The chart is courtesy of the Dead Sea Works.
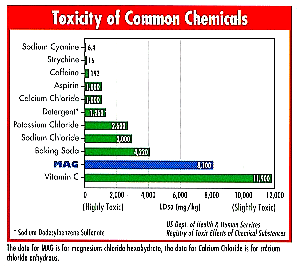 |
Author's Note: In 2001, the USDA published an article entitled "Magnesium Chloride as a De-Icing Agent" that stated magnesium chloride led to "corrosion of steel and aluminum poles and pole hardware." Although used as an effective deicer, updated evidence after the publishing of this article on AsktheBuilder.com shows it can lead to harm of electric utilities. The article can be read here.
Also, a February 2003 Board of Montana Flathead County Commissioners minutes document called "Magnesium Chloride on Roads" on Corrosion-Doctors.org attests to magnesium chloride's corrosiveness on aluminum and steel. In a corrosion comparison done by Colorado D.O.T. and the University of Colorado, it was found that "road salt is more corrosive to the metals than mag chloride on a one time exposure." However, magnesium chloride corrodes over longer periods of time and Greg Fulton, president of the Colorado Motor Carriers Association, said it corroded aluminum on his vehicles as well as led to wiring failure. This document can be read here.
Related Articles: Guidelines for Strong Concrete, Deicing Salt and Concrete, Magnesium Chloride & Deicing Salt Companies
Tim built custom homes and did ultra-custom remodeling for 20+ years before launching his Ask the Builder media presence. He was selected as one of the Big 50 by Remodeling magazine in 1993. This award gave birth to Ask the Builder in October 1993. Tim also operates https://DrawPlumbingPlans.com. His @askthebuilder YouTube channel was the first home improvement channel on YouTube.
I'm a bridge engineer, so I deal with the effects of deicers on concrete and steel on a daily basis.
All chlorides corrode metals when moisture is present. Calcium chloride is especially bad because it's hygroscopic - it will pull water right out of the air (which is why it's used for dust control on dirt roads).
Sodium chloride and calcium chloride don't have much effect on properly cured concrete (but they will corrode any metal reinforcing). However, magnesium chloride (or calcium magnesium acetates) attack concrete directly by a chemical reaction. The main cementitious component in portland cement concrete is calcium silicate hydrate (CSH). The magnesium ions in magnesium chloride replace the calcium ions in CSH to form magnesium silicate hydrate, which is not cemetitious. As you might imagine, when your concrete has nothing cementing it together anymore, that's a problem! So, magnesium chloride should not be used on concrete, and it may also be a problem when it contacts vehicles.
Check out this story: http://ctmirror.org/2015/02/10/snow-plus-salt-equals-connecticut-controversy/